Enzymes are biological catalysts that allow a host of chemical reactions to take place in organisms. They can also be used in synthetic chemistry to perform difficult reactions, e.g., forming carbon-carbon bonds. Enzymes (and more generally, all catalysts) do this by lowering the activation energy of a reaction and stabilizing the transition state. Where does the word enzyme come from?
Enzyme
Origin: Greek, en– (in) + zyme (leavened)
Something leavened in
Louis Pasteur (Pasteur pipettes, pasteurization, etc.), a titanic French chemist of the 1800s, was studying the fermentation of alcohol by yeast. He postulated that the agent responsible for fermentation was a “vital force,” and that only living organisms could perform the task. Wilhelm Kühne coined the term “enzyme” to refer to the process, but it was Eduard Büchner (Büchner funnel) who showed that cell-free yeast extracts could still ferment sugar into alcohol. For this, Büchner received the 1907 Nobel Prize in Chemistry. He named the agent responsible zymase.
Left: Pasteur; Center: Kühne; Right: Büchner
Zymase
Origin: Greek, zyme (leavened) + –ase (enzyme suffix)
That which cleaves “leavened,” i.e., the substrate is from the yeast
Following Büchner’s convention, we use the suffix –ase to denote the substrate of an enzyme, e.g., an enzyme that catalyzes a reaction involving a peroxide is called a peroxidase. Note that this convention is not used for all enzymes. Here are some enzymes with interesting etymologies that are important for digestion:
Amylase
Origin: Greek, a– (without) + myle (mill) + –ase
That which cleaves something unmilled, i.e., starch
Pepsin
Origin: Greek, peptein (to digest)
That which cleaves proteins into peptides
Lipase
Origin: Greek, lipos (fat) + –ase
That which cleaves fats, e.g., triglycerides
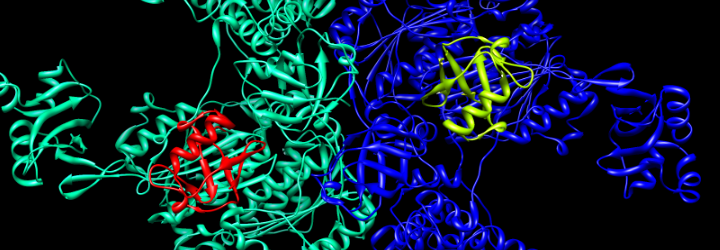
Some enzymes require organic or inorganic cofactors in order to function. The organic cofactors can either be prosthetic groups, which are covalently bound to the enzyme, or they can be coenzymes, which are cleaved from the active site during the reaction. Some cofactors may be involved in allosteric modulation; here, an ion or molecule binds to an area away from the enzyme’s active site to modulate its activity.
Biotin (left) is a cofactor of pyruvate carboxylase (right)
Allosteric
Origin: Greek, allos (other) + stereos (solid, taken to mean 3D shape/position)
Pertaining to another space/position
Are there any cool enzymes with puzzling etymologies that you can think of? Leave a comment below and let’s explore!